May 1131, 11018
Authors
Alekseyenko AA†, Walsh EM†, Wang X†, Grayson AR, Hsi PT, Kharchenko PV, Kuroda MI, and French CA
Genes Dev 2015, doi:10.1101/gad.267583.115.
Abstract
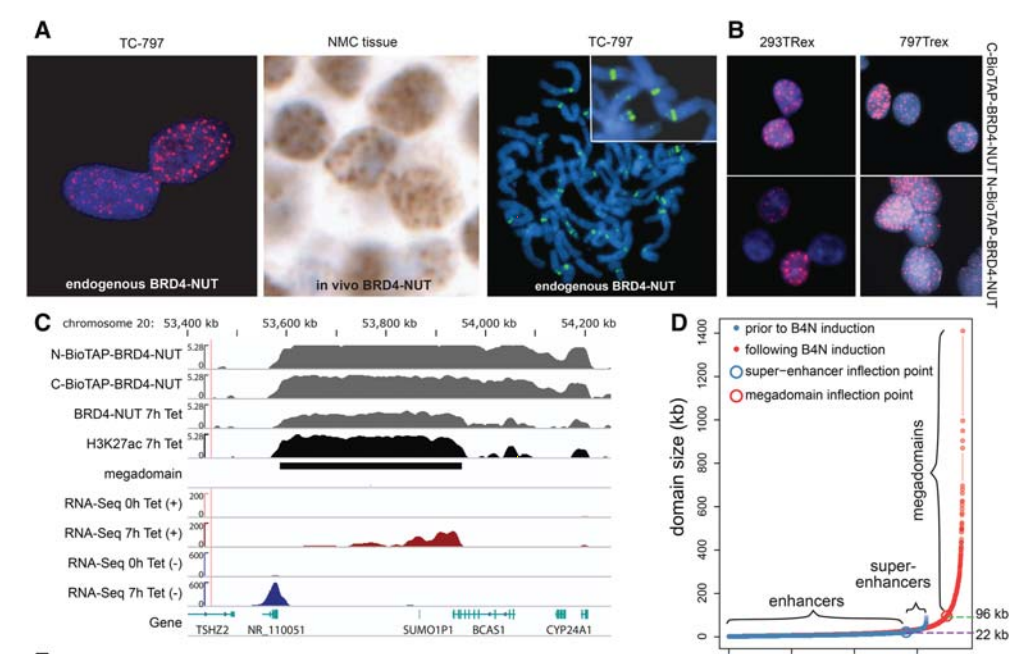
NUT midline carcinoma (NMC), a subtype of squamous cell cancer, is one of the most aggressive human solid malignancies known. NMC is driven by the creation of a translocation oncoprotein, BRD4-NUT, which blocks differentiation and drives growth of NMC cells. BRD4-NUT forms distinctive nuclear foci in patient tumors, which we found correlate with ∼100 unprecedented, hyperacetylated expanses of chromatin that reach up to 2 Mb in size.
These “megadomains” appear to be the result of aberrant, feed-forward loops of acetylation and binding of acetylated histones that drive transcription of underlying DNA in NMC patient cells and naïve cells induced to express BRD4-NUT. Megadomain locations are typically cell lineage-specific; however, the cMYC and TP63 regions are targeted in all NMCs tested and play functional roles in tumor growth. Megadomains appear to originate from select pre-existing enhancers that progressively broaden but are ultimately delimited by topologically associating domain (TAD) boundaries. Therefore, our findings establish a basis for understanding the powerful role played by large-scale chromatin organization in normal and aberrant lineage-specific gene transcription.
Keyword
BRD4; chromatin hyperacetylation; topological domains